Traceability – Transparency to a Safer Supply Chain

Nogales, AZ- Across the industry, providers of fresh fruits and vegetables are responsible for meeting certain criteria when it comes to food safety and traceability record keeping. The process of being able to trace product from the farm to the final consumer is not only considered a best practice, but an essential necessity to preventing or handling a potential foodborne illness. Growers and first receivers who have the ability to properly document and have methods for recording data in regards to harvest shipments are more likely to recover the product faster and more efficiently.
Recently, the FDA has been proposing additional traceability requirements for companies who manufacture, process, grow, handle, or store product which is included on the Food Traceability List. The proposed rule, “Requirements for Additional Traceability Records for Certain Foods” is considered a key component to the FDA’s New Era of Smarter Food Safety Blueprint which is an implementation of the Food Safety Modernization Act’s (FSMA) section 204 (d).
“The proposed requirements would help the FDA rapidly and effectively identify recipients of those foods to prevent or mitigate foodborne illness outbreaks and address credible threats of serious adverse health consequences or death”, per the FDA website.
At the center of this proposal, the FDA as established a Food Traceability List (FTL) (Table 1.0) which requires those who grow, manufacture, process, store/or hold to develop and maintain records know as Key Data Elements (KDEs) which are also associated to Critical Tracking Events (CTEs).
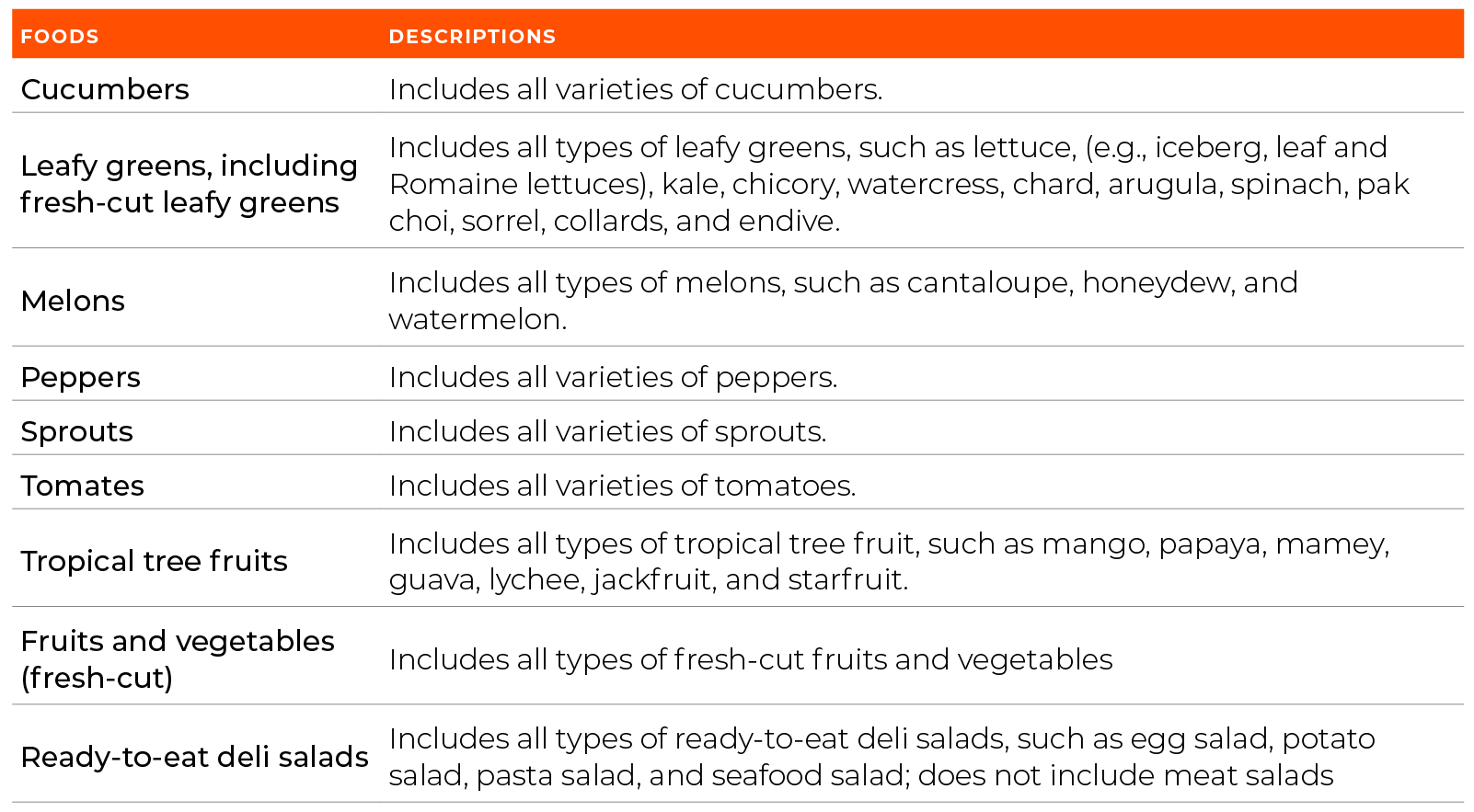
*This list has been simplified to reflect produce items. Other products from FTL not included on this table.
WHAT DOES THIS MEAN FOR DIVINE FLAVOR AND THE GROWERS?

1. CRITICAL TRACKING EVENTS (CTE)
Growing- For fresh fruits and vegetables, growing is the first step of the process. Growers must have systems or Key Data Elements (KDEs) in place for tracking product from their harvest. This generally refers to harvest tags, manifest, and other records related to where the product came from at the farm.
First Receiver- Once the produce is received at the Divine Flavor distribution center, this is considered the first receiving point of the process from the grower to the retailer and final consumer. KDEs and Critical Tracking Events (CTEs) are a very important aspect to the proposed regulation, but this is an area already implemented and is currently being followed by Divine Flavor.
2. TRACEABILITY PROGRAM RECORDS
In addition to the KDEs discussed above, the proposed rule would also require companies who grow, manufacture, process, etc. to have traceability program records. These records are intended to help regulators track and trace products more efficiently. The traceability program would include the following:
A description of records- Bills of lading (BOLs), purchase orders, production logs, etc.
A list of foods from the FTL list- This information would be included in the records and traceability program.
A description of how traceability lot codes are assigned- At Divine Flavor, all products are assigned lot codes which allows products to be uniquely identified to specific farms locations and other useful harvest information.
3.ADDITIONAL REQUIREMENTS
- Records must be maintained (original or electronic)
- Records must be available and sent to the FDA within 24 hours of being requested
- Must be able to assist with the FDA if there are any outbreaks, recall, or any other threat to public health
Currently, the FDA’s proposed rule is currently in the public comment process and has 60 days since the rule was published in the Federal Register. Regardless of the proposed rule, and whether products are exempted from the FTL, Divine Flavor & its growing community believe traceability has always been a key to a safer supply chain and remain committed to implementing the best traceability systems.
Take a look at some past stories regarding traceability:
Resources:
For more information, please contact:
Michael DuPuis
Public Relations Coordinator
+1 (520)-281-8328
mdupuis@divineflavor.com









